
It is because the atoms plus molecules of the substances start rearranging themselves to form a new product. In other words, chemical changes do alter the properties of a substance throughout the reaction. Thus, there is a change in the composition of the material forming a new product. For instance, when you burn a piece of paper or wood, it will change into ash. It results in a new product after the reaction. Definition of Chemical ChangeĪ chemical change is the exact opposite of physical change. 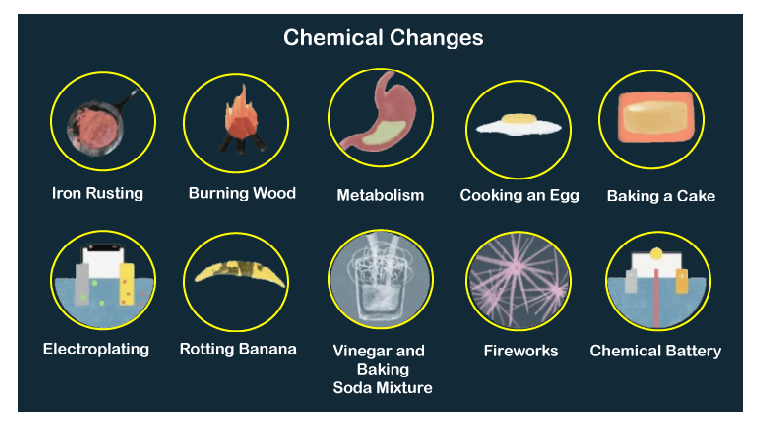
Moreover, the changes are usually reversible and temporary. A physical change does not bring any alternation of any kind on the chemical properties. Thus, this will help you in understanding the difference between physical and chemical change better. Similarly, when water turns into ice or vapour, the chemical properties remain the same, only the physical state changes. However, the properties will be the same. For instance, when you tear a paper, it will only change its shape and size. In here, the chemical properties do not change. Physical change is the kind of change where the matter switches into another form. Whereas, a chemical change is when the internal structure gets affected by a substance and forms a new substance. Moreover, it does not make any internal changes. Physical change is when the physical traits of a substance changes. Thus, in this article, we will study about the difference between physical and chemical change. All of these changes are physical changes or chemical changes. For instance, melting of wax, rusting of iron, bread turning into toast and more. The most definitive way to confirm a chemical reaction is to analyze the chemical compositions of the reactants and products.We encounter a lot of changes in our surroundings day to day that which we may not necessarily notice. An example of a precipitate is yellow cadmium sulfide, which is produced when water solutions of ammonium sulfide and cadmium nitrate are combined. Precipitate FormationĪ precipitate is a solid product that forms as a result of a chemical reaction between liquid reactants. An example of this is when baking soda and vinegar react violently to produce carbon dioxide gas in the famous volcano experiment. When substances react and bubbles or a vapor appears, this is an indication that a chemical reaction is occurring. Change in ColorĪ change in color is a possible indication of a chemical reaction. But it is a strong argument that one has occurred. Because many physical changes also involve heat or light, this is not concrete, definitive evidence of a reaction. One example of this is a fire or spark, which is evidence of combustion. If a reaction emits heat or light, that is an indication that energy has been released from the reaction. So how can we tell if a chemical change has occurred? 1. Indications of a Chemical Reactionīased on its definition, a chemical reaction occurs when a substance undergoes a chemical change. While some physical changes are easily reversible, such as re-freezing melted ice, reversing a chemical change requires another chemical reaction. This process changes the chemical composition of the original substance. When an iron nail comes into contact with water and is then exposed to the air, it rusts, forming a brown-red substance. Once melted or boiled, the water may be in a different form (solid ice or gaseous water vapor), but it is still water, H 2O, and it still has the same chemical composition.Ī chemical change happens when a substance’s chemical identity changes. For example, freezing or boiling water is a physical change. In a physical change, a compound’s shape may change, but its chemical identity will not. Let’s looks at some chemical change examples, and chemical reaction examples. Chemical equations express chemical reactions in terms of symbols and chemical formulas of the compounds involved in reactions. 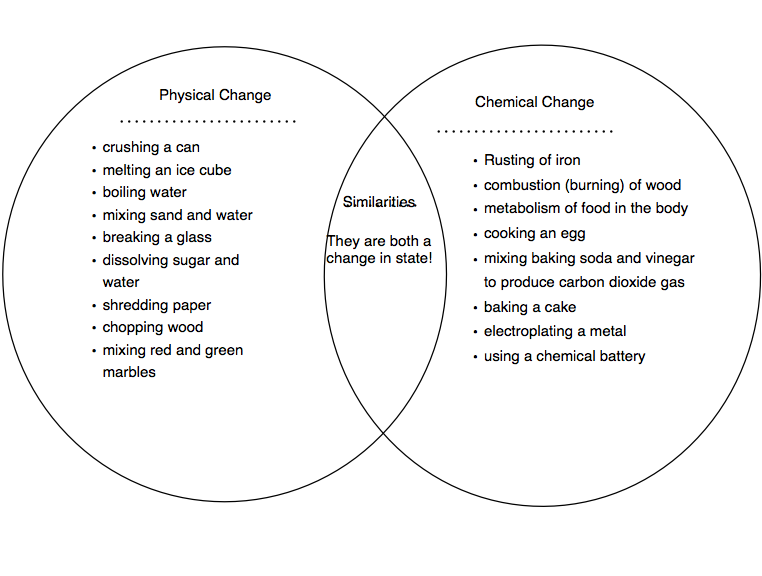
So what is a chemical reaction? What is a chemical change, and what is a physical change? A chemical reaction occurs when compounds or substances undergo a chemical change to form different compounds or substances.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
